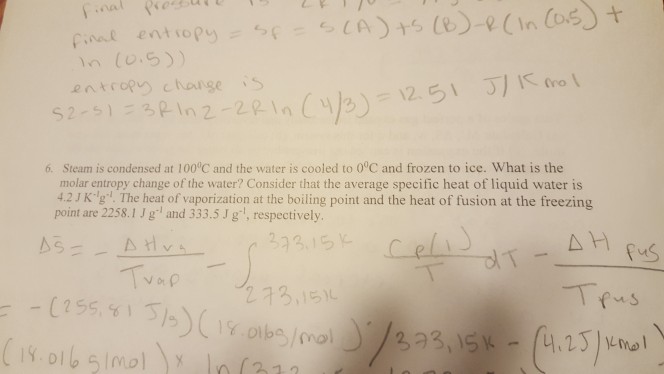Show transcribed image text (0.5) entrop change S 6. Steam is condensed at 100°C and the water is cooled to 0°C and frozen to ice. What is the molar entropy change of the water? Consider that the average specific heat of liquid water is 4.2J Kg. The heat of vaporization at the boiling point and the heat of fusion at the freezing point are 2258.1 J g and 333.5 J gl, respectively. 273,15% (23)wdl
(0.5) entrop change S 6. Steam is condensed at 100°C and the water is cooled to 0°C and frozen to ice. What is the molar entropy change of the water? Consider that the average specific heat of liquid water is 4.2J Kg. The heat of vaporization at the boiling point and the heat of fusion at the freezing point are 2258.1 J g and 333.5 J gl, respectively. 273,15% (23)wdl