
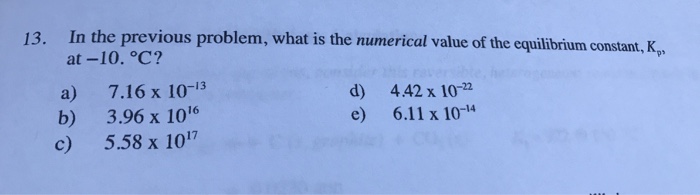
Show transcribed image text Consider the heterogeneous reaction below: Cl2(H2O), (s) ←→ C, (g) + 8H20 (g) When solid Cu(HO, is placed in a 20 0 L container at -10. C, the equilibrium partial pressure of water vapor produced is 5.34 x 10 atm. What is the equilibrium partial pressure of chlorine gas, Clh? pay attention to physical states. a) 9.22 x 10-3 atm b) 0.542 atm c) 6.68 x 10- atm 12. Hints: Use an ICE table and d) 4.91 x 10-2 atm e) 1.13 x 10 3 atm
Consider the heterogeneous reaction below: Cl2(H2O), (s) ←→ C, (g) + 8H20 (g) When solid Cu(HO, is placed in a 20 0 L container at -10. C, the equilibrium partial pressure of water vapor produced is 5.34 x 10 atm. What is the equilibrium partial pressure of chlorine gas, Clh? pay attention to physical states. a) 9.22 x 10-3 atm b) 0.542 atm c) 6.68 x 10- atm 12. Hints: Use an ICE table and d) 4.91 x 10-2 atm e) 1.13 x 10 3 atm






