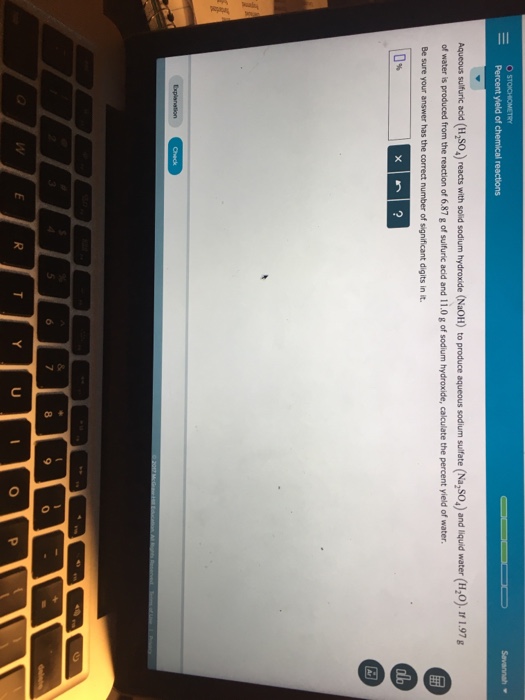
Show transcribed image text Savannah ▼ Percent yield of queous sulfturic acid (H,SO,) reacts with solid sodium hydroxide (NaOHl) to produce aqueous sodium sulfate (Na,SO,) and liquid water (H,O) If 1.97g of water is produced from the reaction of 6.87 g of sulfuric acid and 11.0 g of sodium hydroxide, calculate the percent yield of water. Be sure your answer has the correct number of significant digits in it. 0% 0
Savannah ▼ Percent yield of queous sulfturic acid (H,SO,) reacts with solid sodium hydroxide (NaOHl) to produce aqueous sodium sulfate (Na,SO,) and liquid water (H,O) If 1.97g of water is produced from the reaction of 6.87 g of sulfuric acid and 11.0 g of sodium hydroxide, calculate the percent yield of water. Be sure your answer has the correct number of significant digits in it. 0% 0






