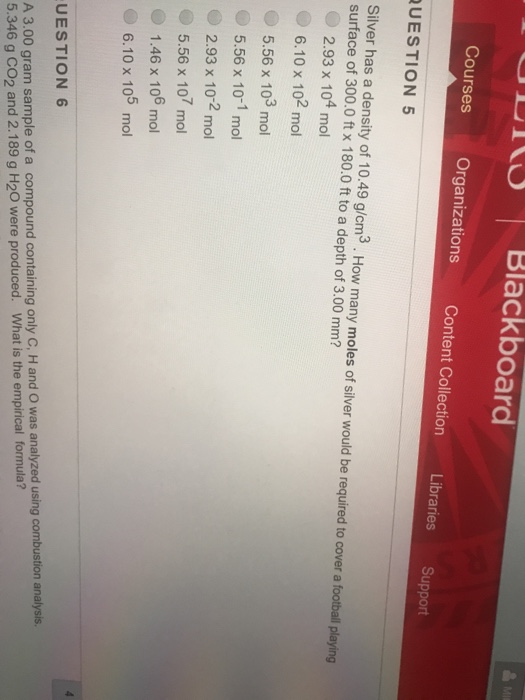
Show transcribed image text Iy Blackboard Courses Organizations Content Collection Libraries Support UESTION 5 Silver has a density of 10.49 g/cm3. How many moles of silver would be required to cover a football playing surface of 300.0 ft x 180.0 ft to a depth of 3.00 mm? 2.93 x 104 mol 6.10 x 102 mol 5.56 x 103 mol 5.56 x 10-1 mol 2.93 x 102 mo 5.56 x 107 mo 1.46 x 106 mol 6.10 x 105 mol A 3.00 gram sample of a compound containing only C, H and O was analyzed using combustion analysis 5.346 g CO2 and 2.189 g H20 were produced. What is the empirical formula? UESTION 6
Iy Blackboard Courses Organizations Content Collection Libraries Support UESTION 5 Silver has a density of 10.49 g/cm3. How many moles of silver would be required to cover a football playing surface of 300.0 ft x 180.0 ft to a depth of 3.00 mm? 2.93 x 104 mol 6.10 x 102 mol 5.56 x 103 mol 5.56 x 10-1 mol 2.93 x 102 mo 5.56 x 107 mo 1.46 x 106 mol 6.10 x 105 mol A 3.00 gram sample of a compound containing only C, H and O was analyzed using combustion analysis 5.346 g CO2 and 2.189 g H20 were produced. What is the empirical formula? UESTION 6






